Категория: Новости.
Dalton Transactions Issue 12, Pages 3895-3905
Anastasia I. Solomatina, Irina O. Aleksandrova, Antti J. Karttunen, Sergey P. Tunik, Igor O. Koshevoy
Dibenzothiophene-platinated complexes: probing the effect of ancillary ligands on the photophysical performance
Dalton Transactions Issue 12, Pages 3895-3905
DOI: 10.1039/C7DT00349H
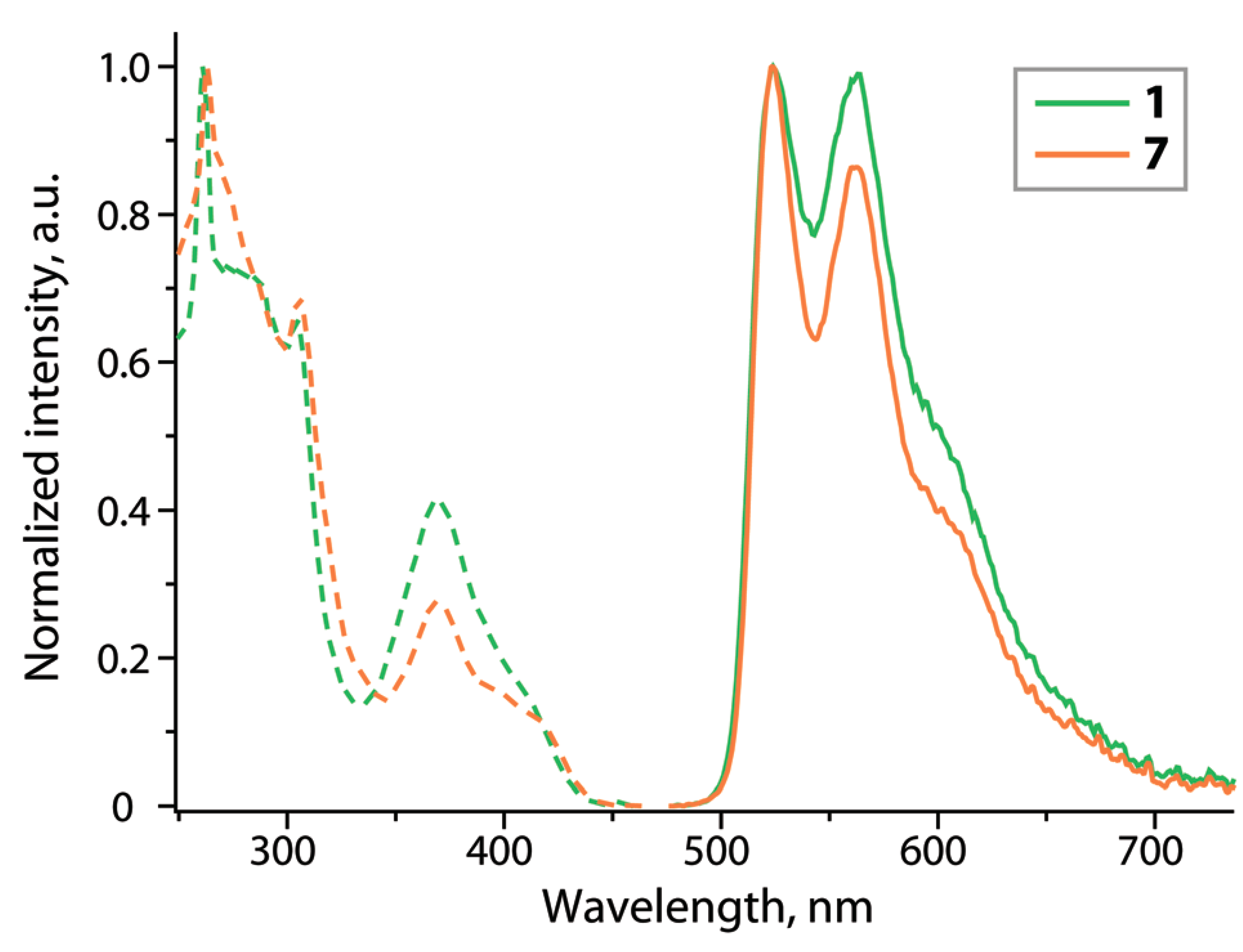
Cyclometalation of dibenzothienyl-pyridine (HPyDBT) afforded a series of platinum(II) complexes Pt(PyDBT)(L)Cl (L = DMSO, 1; P(p-C6H4-X)3 (X = H, 2; CF3, 3; OMe, 4; NPh2, 5); 1,3,5-triaza-7-phosphaadamantane, 6; 2,6-dimethylphenyl isocyanide, 7). Chelating bidentate LL ligands formed cationic compounds [Pt(PyDBT)(LL)]+ (LL = 1,2-bis(diphenylphosphino)benzene, 8; 2,2’-bipyridine, 9; 1,10-phenanthroline, 10). Oxidation of a thienyl sulfur atom allowed for the isolation of the sulfone derivative Pt(PyDBT)(PPh3)Cl (11). The title complexes were characterized crystallographically (except 7). Investigation of their photophysical behavior revealed solid state phosphorescence with quantum yields up to 0.45 for neat powders. The ancillary ligands L show a minor influence on the emission energies of the neutral compounds, but affect dramatically the intensity of luminescence. In contrast, the cationic species with diimine ligands demonstrate a significant contribution of the LL fragments into the emissive T1 states that leads to a certain mixing of 3IL and 3LL’CT transitions and causes a substantial bathochromic shift of emission.
Категория: Новости.
Journal of Molecular Liquids Volume 230, March 2017, Pages 113–120
V.S. Fundamensky, T.A. Kochina, Y.A. Kondratenko, A.A. Zolotarev, Yu.G. Vlasov, I.S. Ignatyev
Ionic liquids based on triethanolammonium salts of dicarboxylic acids (oxalic, malonic, succinic). Crystal structure and cation-anion interaction
Journal of Molecular Liquids Volume 230, March 2017, Pages 113–120
DOI: 10.1016/j.molliq.2016.12.111
The series of ionic liquids based on triethanolammonium salts of dicarboxylic acids (oxalic, malonic and succinic) was synthesized, characterized by IR and H1, C13 NMR spectroscopy, thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC). Their structure was determined by single-crystal X-ray diffraction. It was found that triethanolammonium [(HOCH2CH2)3NH]+ cations in salts of oxalic (1), malonic (2) and succinic acids (3) containing monoanions [OOC(CH2)nCOOH]- (n=0-2) have the endo conformation and the ammonium proton (Ham) resides inside the “lampshade” formed by three CH2CH2OH branches connected with nitrogen atom. The asymmetric cell of the second modification of the succinic acid salt (4) includes two triethanolammonium cations and a succinate dianion [(HOCH2CH2)3NH]2+[OOC(CH2)2COO]2-. In this salt (4) one CH2CH2OH branch of the triethanolammonium cation is rotated around the N-C bond (endo-exo conformation) and forms infinite TEA chains. This pattern was firstly found in organic salts of TEA. The obtained results show that different structures of triethanolammonium cations in the salts of dicarboxylic acids have significant influence on the cation-anion interaction.
Категория: Новости.
Glass Physics and Chemistry, 2017, Vol. 43, No. 4, pp. 298–301
A. A. Razumtsev, Yu. S. Tver’yanovich, Fahd S. Khan, I. E. Kolesnikov, and A. V. Kurochkin
«Spectral Properties of Glass (15Ga2S3 · 85GeS2) Doped with Erbium»
Glass Physics and Chemistry, 2017, Vol. 43, No. 4, pp. 298–301
DOI: 10.1016/j.jnoncrysol.2016.11.038
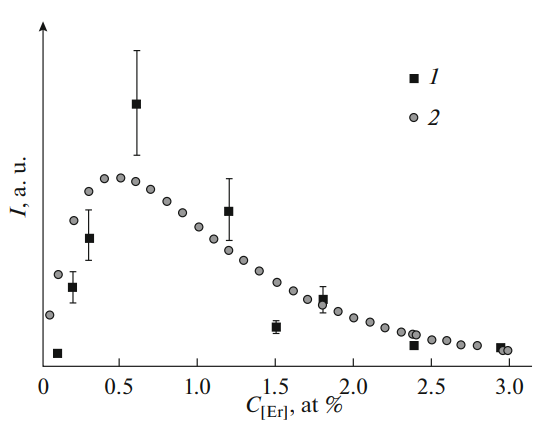
For chalcogenide glasses in the system (1 – x)[0.15Ga2S3 · 0.85GeS2] · xEr2S3, the absorption and luminescence spectra are investigated and the X-ray diffraction analysis is performed. A small shift in the position of the erbium absorption band with the increase of its content in the glass indicates the decrease of the effective charge on it, while the negligible changes in the angle position of the first sharp diffraction peak points to the constancy of the glass’s intermediate-order parameter. The possibility of describing the dependence of the intensity of erbium luminescence on its concentration using the earlier suggested equation has been discussed.
Категория: Новости.
17.03.2017 в РЦ ОЛМИВ состоялся визит сотрудников отдела научно-технической экспертизы Государственного Эрмитажа: Гавриленко Л.С.Ю Григорьева И.А., Чугунова К.С. и Одриковская А.Ю. Обсуждались вопросы научно-технического сотрудничества.

Категория: Новости.
Journal of Structural Chemistry. Vol. 58, No. 2, March 2017, pp. 399-405
S. V. Paston, A. M. Polyanichko, O. V. Shulenina
STUDY OF DNA INTERACTIONS WITH Cu2+ AND Mg2+ IONS IN THE PRESENCE OF CAFFEINE
Journal of Structural Chemistry. Vol. 58, No. 2, March 2017, pp. 399-405
DOI: 10.1134/S0022476617020263
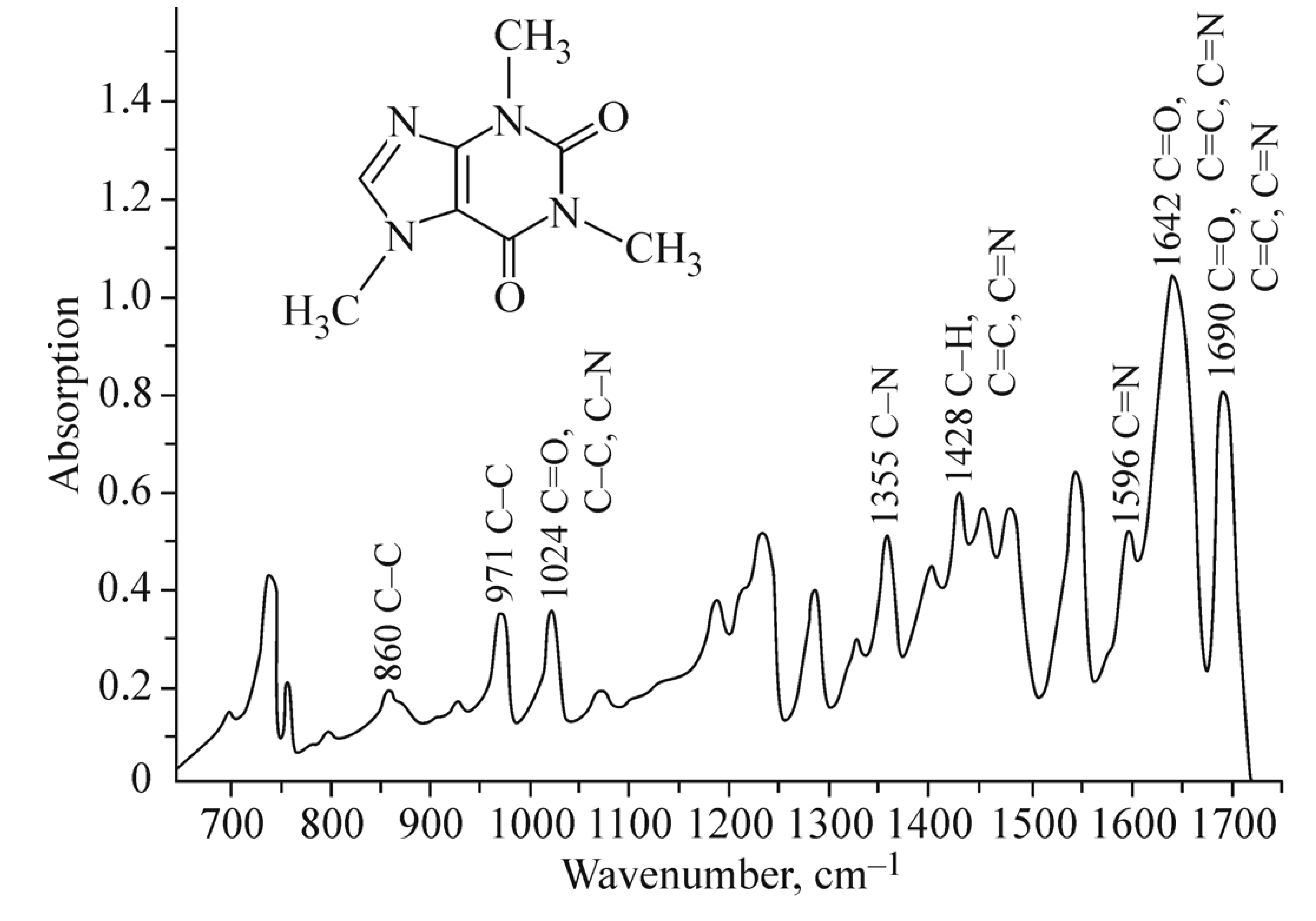
A study of caffeine effects on the complexation of the DNA molecule with Cu2+ and Mg2+ ions in solution is presented. DNA binding with cations and caffeine is traced from changes in DNA circular dichroism and UV absorption spectra. The complexation of caffeine with M2+ ions is studied by IR spectroscopy. Caffeine is found to form complexes of different structure with Mg2+ and Cu2+ ions in solution. In the presence of caffeine, DNA solutions with MCl2 show signs of formation of intricate complexes involving nitrogenous bases. In DNA solutions with CuCl2, the presence of caffeine causes additional destabilization of the secondary DNA structure. In DNA solutions with MgCl2, the presence of caffeine leads to the formation of new complexes with the participation of Mg2+, caffeine, and bases without disrupting the secondary DNA structure.

 Русский (РФ)
Русский (РФ)  English (UK)
English (UK) 


