Posted in Новости
On April 24, a joint seminar of the RC OLMIV, the Department of Laser Chemistry and Materials Science and representatives of the ISC RAS named after Grebenshchikov took place in the lecture hall of RC OLMIV.
The seminar lectures:
O.A. Shilova, prof., Institute of Silicate Chemistry named after. Grebenshchikov and VN Shchelushkin, head of the technological group, company group Solex, Ryazan.
Issues of joint research in the field of research of new materials, in particular, for solar energy were discussed.
Posted in Новости
Polyhedron Volume 130, Pages 176-183
Y. Kondratenko, V. Fundamenskya, I. Ignatyev, A. Zolotarev, T. Kochina, V. Ugolkov
Synthesis and crystal structure of two zinc-containing complexes of triethanolamine
Polyhedron Volume 130, Pages 176-183
DOI: 10.1016/j.poly.2017.04.022
Two new zinc-containing complexes [Zn2(TEA)(C6H5COO)3] (1) and ([Zn(TEA)(H2O)2]SO4)·H2O (2) were synthesized and characterized by IR spectroscopy, elemental analysis, DSC and TG analysis. Their structure was determined by single-crystal X-ray diffraction. In the binuclear, mixed-ligand [Zn2(TEA)(C6H5COO)3] complex, one zinc atom is six-coordinated by nitrogen and three oxygen atoms of tetradentate triethanolamine (TEA) and two oxygen atoms of two different benzoate ligands, forming the distorted octahedron of the ZnNO5 type. The second zinc atom is five-coordinated, forming the distorted trigonal bipyramid. Two zinc atoms are bridged by two carboxylate groups of two benzoate ligands and one oxygen atom of the deprotonated hydroxyethyl group of TEA. Cationic complex 2 consists of [Zn(TEA)(H2O)2]2+ cations and SO42- anions. The coordination polyhedron around the zinc atom corresponds to a distorted octahedron (ZnNO5 type). The TEA ligand is tetradentately coordinated to the cation, forming three chelate rings. The coordination sphere of the Zn cation is completed by two aqua ligands.
Posted in Новости
REVIEWS ON ADVANCED MATERIALS SCIENCE Volume 47, No. 1, Pages 71-77
O.Yu. Kurapova, V.G. Konakov, A.S. Grashchenko, N.N. Novik, S.N. Golubev and I.A. Ovid’ko
NANOTWINNED COPPER-GRAPHENE COMPOSITES WITH HIGH HARDNESS
REVIEWS ON ADVANCED MATERIALS SCIENCE Volume 47, No. 1, Pages 71-77
This paper addresses fabrication, structure, and hardness characteristics of nanotwinned (ntw) copper-graphene composites. The composites were fabricated by electrodeposition from 1M CuSO4·6H2O mixed water-alcohol solution containing graphene-graphite mixture stabilized by non-ionic surfactant. We fabricated two-layer solids each consisting of ntw copper layer and ntw copper-graphene layer. The synthesized two-layer specimens were examined in nanoindentation tests and showed high hardness values up to 3 GPa. The maximum hardness value of 3 GPa is higher than those of pure ntw copper and copper-graphene composites, taken from the literature.
Posted in Новости
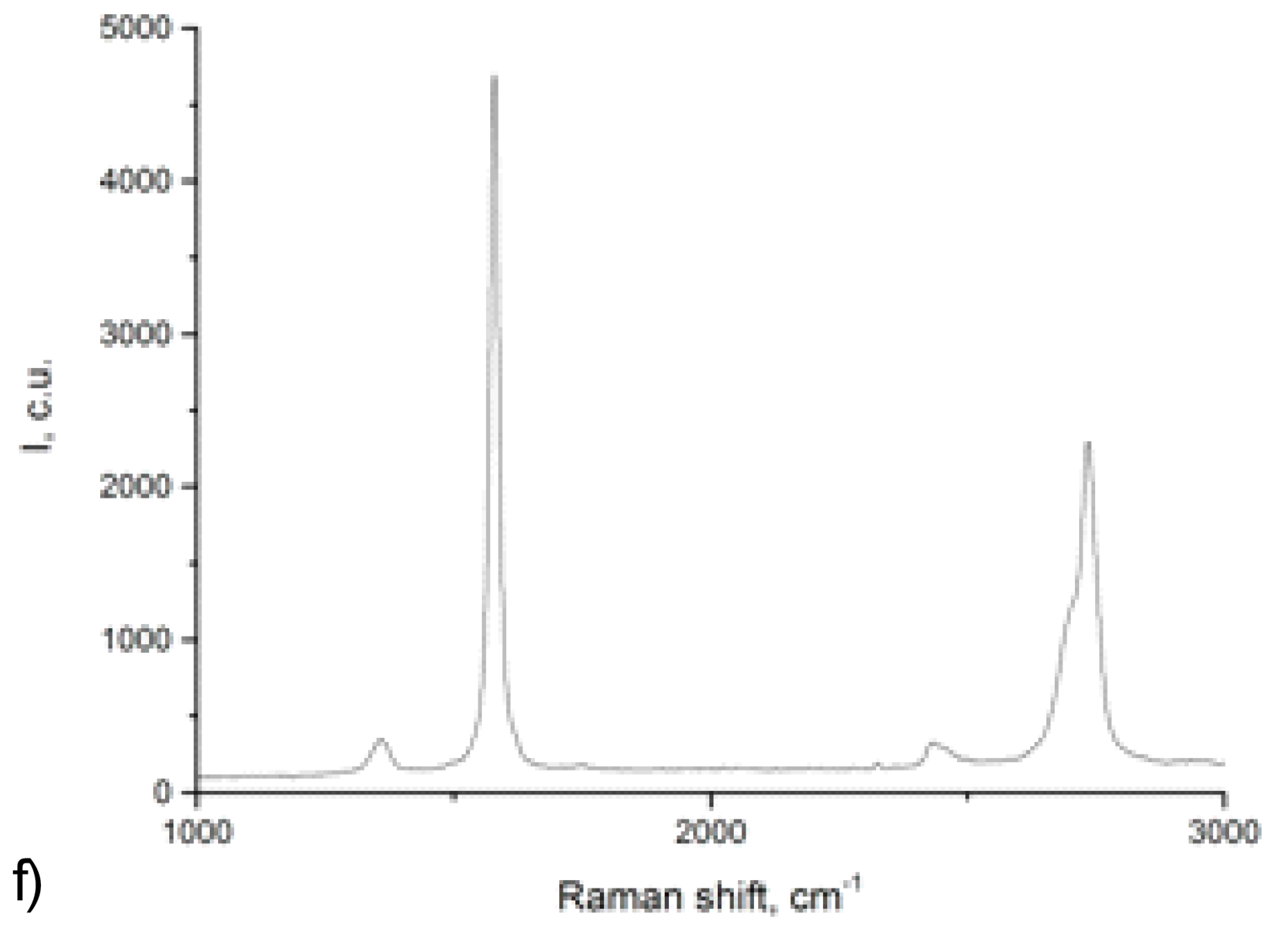
Dalton Transactions Issue 12, Pages 3895-3905
Anastasia I. Solomatina, Irina O. Aleksandrova, Antti J. Karttunen, Sergey P. Tunik, Igor O. Koshevoy
Dibenzothiophene-platinated complexes: probing the effect of ancillary ligands on the photophysical performance
Dalton Transactions Issue 12, Pages 3895-3905
DOI: 10.1039/C7DT00349H
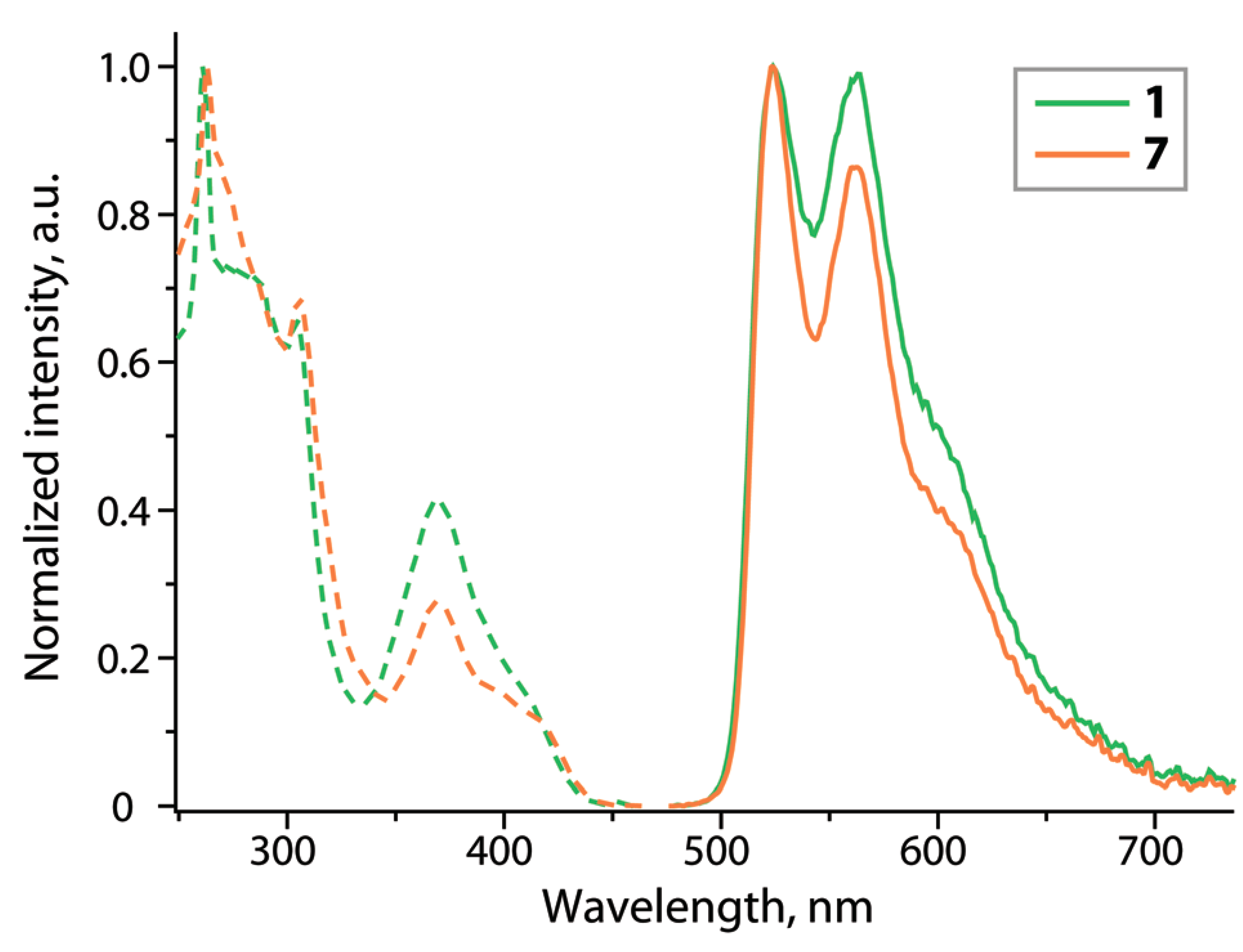
Cyclometalation of dibenzothienyl-pyridine (HPyDBT) afforded a series of platinum(II) complexes Pt(PyDBT)(L)Cl (L = DMSO, 1; P(p-C6H4-X)3 (X = H, 2; CF3, 3; OMe, 4; NPh2, 5); 1,3,5-triaza-7-phosphaadamantane, 6; 2,6-dimethylphenyl isocyanide, 7). Chelating bidentate LL ligands formed cationic compounds [Pt(PyDBT)(LL)]+ (LL = 1,2-bis(diphenylphosphino)benzene, 8; 2,2’-bipyridine, 9; 1,10-phenanthroline, 10). Oxidation of a thienyl sulfur atom allowed for the isolation of the sulfone derivative Pt(PyDBT)(PPh3)Cl (11). The title complexes were characterized crystallographically (except 7). Investigation of their photophysical behavior revealed solid state phosphorescence with quantum yields up to 0.45 for neat powders. The ancillary ligands L show a minor influence on the emission energies of the neutral compounds, but affect dramatically the intensity of luminescence. In contrast, the cationic species with diimine ligands demonstrate a significant contribution of the LL fragments into the emissive T1 states that leads to a certain mixing of 3IL and 3LL’CT transitions and causes a substantial bathochromic shift of emission.
Posted in Новости
Journal of Molecular Liquids Volume 230, March 2017, Pages 113–120
V.S. Fundamensky, T.A. Kochina, Y.A. Kondratenko, A.A. Zolotarev, Yu.G. Vlasov, I.S. Ignatyev
Ionic liquids based on triethanolammonium salts of dicarboxylic acids (oxalic, malonic, succinic). Crystal structure and cation-anion interaction
Journal of Molecular Liquids Volume 230, March 2017, Pages 113–120
DOI: 10.1016/j.molliq.2016.12.111
The series of ionic liquids based on triethanolammonium salts of dicarboxylic acids (oxalic, malonic and succinic) was synthesized, characterized by IR and H1, C13 NMR spectroscopy, thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC). Their structure was determined by single-crystal X-ray diffraction. It was found that triethanolammonium [(HOCH2CH2)3NH]+ cations in salts of oxalic (1), malonic (2) and succinic acids (3) containing monoanions [OOC(CH2)nCOOH]- (n=0-2) have the endo conformation and the ammonium proton (Ham) resides inside the “lampshade” formed by three CH2CH2OH branches connected with nitrogen atom. The asymmetric cell of the second modification of the succinic acid salt (4) includes two triethanolammonium cations and a succinate dianion [(HOCH2CH2)3NH]2+[OOC(CH2)2COO]2-. In this salt (4) one CH2CH2OH branch of the triethanolammonium cation is rotated around the N-C bond (endo-exo conformation) and forms infinite TEA chains. This pattern was firstly found in organic salts of TEA. The obtained results show that different structures of triethanolammonium cations in the salts of dicarboxylic acids have significant influence on the cation-anion interaction.



 English (UK)
English (UK)  Русский (РФ)
Русский (РФ) 
